Michael Jahn, 2022-10-17
Utility functions for bioinformatics work
This package contains utility functions or wrappers for bioinformatics work. It is not intended to be a full grown R package but is maintained as a package for the sake of accessability and documentation. Feel free to copy, fork or source functions that you find useful.
To install the package directly from github, use this function from
devtools package in your R session:
require(devtools)
devtools::install_github("https://github.com/m-jahn/R-tools")Aggregate peptide abundances to protein abundances.
Similar to the openMS module ProteinQuantifier, this function provides different methods to aggregate peptide intensities to their parent proteins. It is mainly intended for the use with (raw) Diffacto results, a table of peptide intensities and covariation scores (weights) that can be used to filter peptides before aggregating them up to protein abundances.
# load additional dependencies
library(Rtools)
# generate data frame
df <- data.frame(
protein = c("A", "B", "C", "C/D", "C/D/E", "E", "F", "G"),
n_protein = c(1,1,1,2,3,1,1,1),
weight = rep(1,8),
peptide = letters[1:8],
ab1 = sample(1:100, 8),
ab2 = sample(1:100, 8),
ab3 = sample(1:100, 8)
)
aggregate_pep(
data = df,
sample_cols = c("ab1", "ab2", "ab3"),
protein_col = "protein",
peptide_col = "peptide",
n_protein_col = "n_protein",
split_ambiguous = TRUE,
split_char = "/",
method = "sum"
)## [1] "ab1" "ab2" "ab3"
## # A tibble: 7 × 5
## protein n_peptides ab1 ab2 ab3
## <chr> <int> <dbl> <dbl> <dbl>
## 1 A 1 31 14 9
## 2 B 1 79 25 93
## 3 C 3 80.3 158. 87.3
## 4 D 2 29.3 68.5 15.3
## 5 E 2 64.3 120 44.3
## 6 F 1 50 57 100
## 7 G 1 43 92 83
Apply normalization based on different published methods. This function
is a wrapper applying different normalization functions from other
packages, such as limma, justvsm and preprocesscore. These are not
imported automatically but have to be installed separately.
Alternatively, it will apply any custom normalization functions that is
passed to the norm_function argument.
df <- data.frame(
protein = LETTERS[1:5],
cond1 = sample(1:100, 5),
cond2 = sample(1:100, 5),
cond3 = sample(1:100, 5)
)
# normalize protein abundance to obtain identical median;
# function borrowed from limma::normalizeMedianValues()
median_norm <- function(x) {
cmed <- log(apply(x, 2, median, na.rm = TRUE))
cmed <- exp(cmed - mean(cmed))
t(t(x)/cmed)
}
df_norm <- apply_norm(
df,
norm_function = median_norm,
sample_cols = 2:ncol(df),
ref_cols = NULL
)
# the data after normalization
print(df_norm)## protein cond1 cond2 cond3
## 1 A 18.58728 39.23981 54.78766
## 2 B 40.27244 83.71160 17.02860
## 3 C 41.82138 18.31191 19.99009
## 4 D 22.20147 23.54389 44.42243
## 5 E 39.23981 107.25548 39.23981
# Has the normalization worked? We can compare column medians
# for original and normalized data
apply(df[2:4], 2, median)## cond1 cond2 cond3
## 76 15 53
apply(df_norm[2:4], 2, median)## cond1 cond2 cond3
## 39.23981 39.23981 39.23981
Cluster levels of a factor based on a response and a grouping variable. The function changes the order of levels of a factor by clustering levels according to similarity of a second response variable, and an optional third grouping variable.
# set seed to obtain same values
set.seed(123)
# a data frame with 5 observations for 5 different groups (A to E)
df <- data.frame(
fc = factor(rep(letters[1:5], 5)),
group = rep(LETTERS[1:5], each = 5),
response = rnorm(25)
)
# levels in alphabetical order
levels(df$fc)## [1] "a" "b" "c" "d" "e"
# reorder levels of "fc" by clustering values in "response" over "groups"
levels(with(df, fct_cluster(fc, group, response)))## [1] "c" "a" "e" "b" "d"
# also works with NA or infinite values;
# infinite values are internally replaced with NA to allow clustering
df[c(1,6,7), "response"] <- -Inf
levels(with(df, fct_cluster(fc, group, response)))## [1] "c" "a" "e" "b" "d"
# missing combinations of variables are completed with NA internally
df <- df[-c(1,6), ]
levels(with(df, fct_cluster(fc, group, response)))## [1] "c" "a" "e" "b" "d"
# different order of factor level does not change result
df$fc <- factor(df$fc, c("c","b","e","d", "a"))
levels(with(df, fct_cluster(fc, group, response)))## [1] "c" "a" "e" "b" "d"
Convenience wrapper to TopGO package (Rahnenfueher et al.). This function carries out a TopGO gene ontology enrichment on a data set with custom protein/gene IDs and GO terms. The function takes as main input a data frame with three specific columns: cluster numbers, Gene IDs, and GO terms. Alternatively, these can also be supplied as three individual lists.
# The get_topgo function will require the TopGO package
# as an additional dependency that is not automatically
# attached with this package.
library(topGO)
# a list of arbitrary GO terms
go_terms <- c(
"GO:0006412", "GO:0015979", "GO:0046148", "GO:1901566", "GO:0042777", "GO:0006614",
"GO:0016114", "GO:0006605", "GO:0090407", "GO:0031564", "GO:0032784", "GO:0052889",
"GO:0032787", "GO:0043953", "GO:0046394", "GO:0042168", "GO:0009124", "GO:0006090",
"GO:0016108", "GO:0016109", "GO:0016116", "GO:0016117", "GO:0065002", "GO:0006779",
"GO:0072330", "GO:0046390", "GO:0006754", "GO:0018298", "GO:0006782", "GO:0022618",
"GO:0042255", "GO:0046501", "GO:0070925", "GO:0071826", "GO:0006783", "GO:0009156"
)
# construct a sample data set with 26 different genes in 2 different groups
# and test which (randomly sampled) GO terms might be enriched in both groups.
# We randomly sample 1 to 3 GO terms per gene. They need to be formatted as one
# string of GO terms separated by "; ".
# set seed to obtain same values
set.seed(123)
df <- data.frame(
GeneID = LETTERS,
cluster = rep(c(1, 2), each = 13),
Gene.ontology.IDs = sapply(1:26,
function(x) paste(sample(go_terms, sample(1:3, 1)), collapse = ";")
),
stringsAsFactors = FALSE
)
# test if GO terms are enriched in group 1 against background
get_topgo(df, selected.cluster = 1, topNodes = 5)## GO.ID Term Annotated Significant
## 1 GO:0044249 cellular biosynthetic process 16 11
## 2 GO:0009058 biosynthetic process 17 11
## 3 GO:1901576 organic substance biosynthetic process 17 11
## 4 GO:0018130 heterocycle biosynthetic process 8 6
## 5 GO:0019438 aromatic compound biosynthetic process 8 6
## Expected classicFisher weightedFisher elimFisher SigGenes
## 1 8.0 0.021 0.45 0.021 A,B,C,D,E,F,I,J,K,L,M
## 2 8.5 0.048 1.00 0.048 A,B,C,D,E,F,I,J,K,L,M
## 3 8.5 0.048 0.30 0.048 A,B,C,D,E,F,I,J,K,L,M
## 4 4.0 0.101 1.00 0.101 C,D,E,J,K,M
## 5 4.0 0.101 1.00 0.101 C,D,E,J,K,M
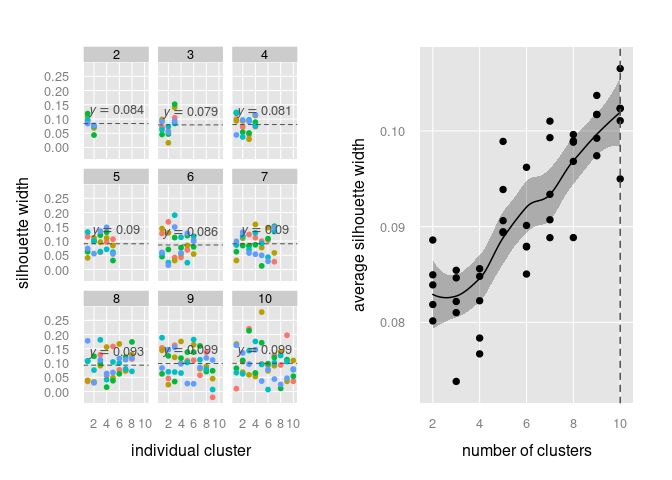
Wrapper function to perform silhouette analysis on different cluster numbers. Silhouette analysis shows the clusters that have explanatory power. That includes clusters that are best separated from the neighbours resulting in a higher average silhoutte width (the decisive metric to judge optimal cluster number). This function applies the silhouette analysis iteratively for a vector of different cluster numbers and stores results in a list.
# generate a random matrix that we use for clustering with the
# format of 100 rows (e.g. determined gene expression) and 10
# columns (conditions)
mat <- matrix(rnorm(1000), ncol = 10)
# we can perform clustering on this matrix using e.g. hclust:
# there is clearly no good separation between different clusters of 'genes'
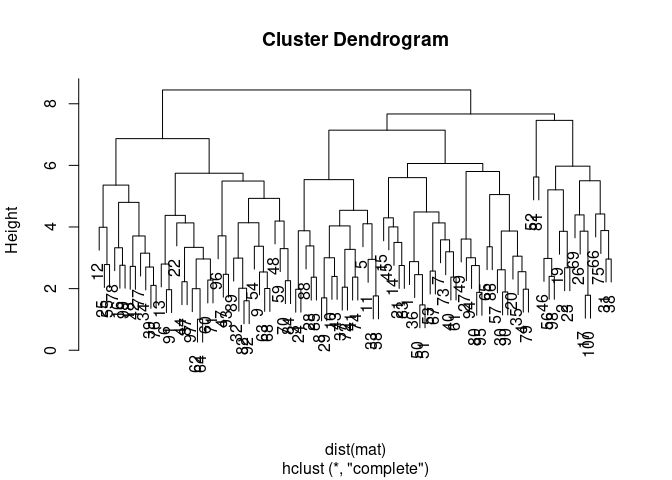
clust <- hclust(dist(mat))
plot(clust)# perform silhouette analysis for 2 to 10 different clusters
sil_result <- silhouette_analysis(mat, n_clusters = 2:10)
# plot results
print(sil_result$plot_clusters, split = c(1,1,2,1), more = TRUE)
print(sil_result$plot_summary, split = c(2,1,2,1))Parse Kegg Brite xml files step-by-step. This script is a small utility to parse Kegg Brite XML files and return a regular data frame instead. The function take no other argument than a data frame. Changes that need to be made to the Kegg XML file before applying the function, e.g. simply using a text editor:
- replace double spaces ’ ’ by tabs ’
- remove first lines until regular content begins
- possibly add some trailing tabs or commas to end of first line (4 to 5) so that read.table knows how many columns to expect
- read raw data frame into R using read.table(“/path/to/file”, fill = TRUE, sep = “, row.names = NULL, stringsAsFactors = FALSE, quote =”“)
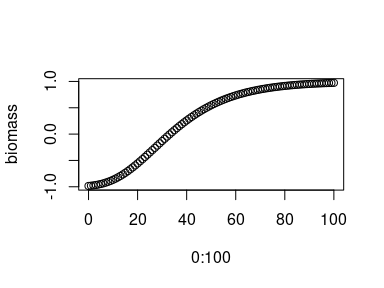
Simulate growth according to the Baranyi growth model.
# simulate growth according to the Baranyi growth model
# for a growth period of 100 hours
biomass <- baranyi_fun(
LOG10N0 = -1, LOG10Nmax = 1,
mumax = 0.1, lag = 10, t = 0:100)
# plot time versus biomass
plot(0:100, biomass)Simulate growth according to the Gompertz modified growth model.
# simulate growth according to the Baranyi growth model
# for a growth period of 100 hours
biomass <- gompertzm_fun(
LOG10N0 = -1, LOG10Nmax = 1,
mumax = 0.1, lag = 10, t = 0:100)
# plot time versus biomass
plot(0:100, biomass)